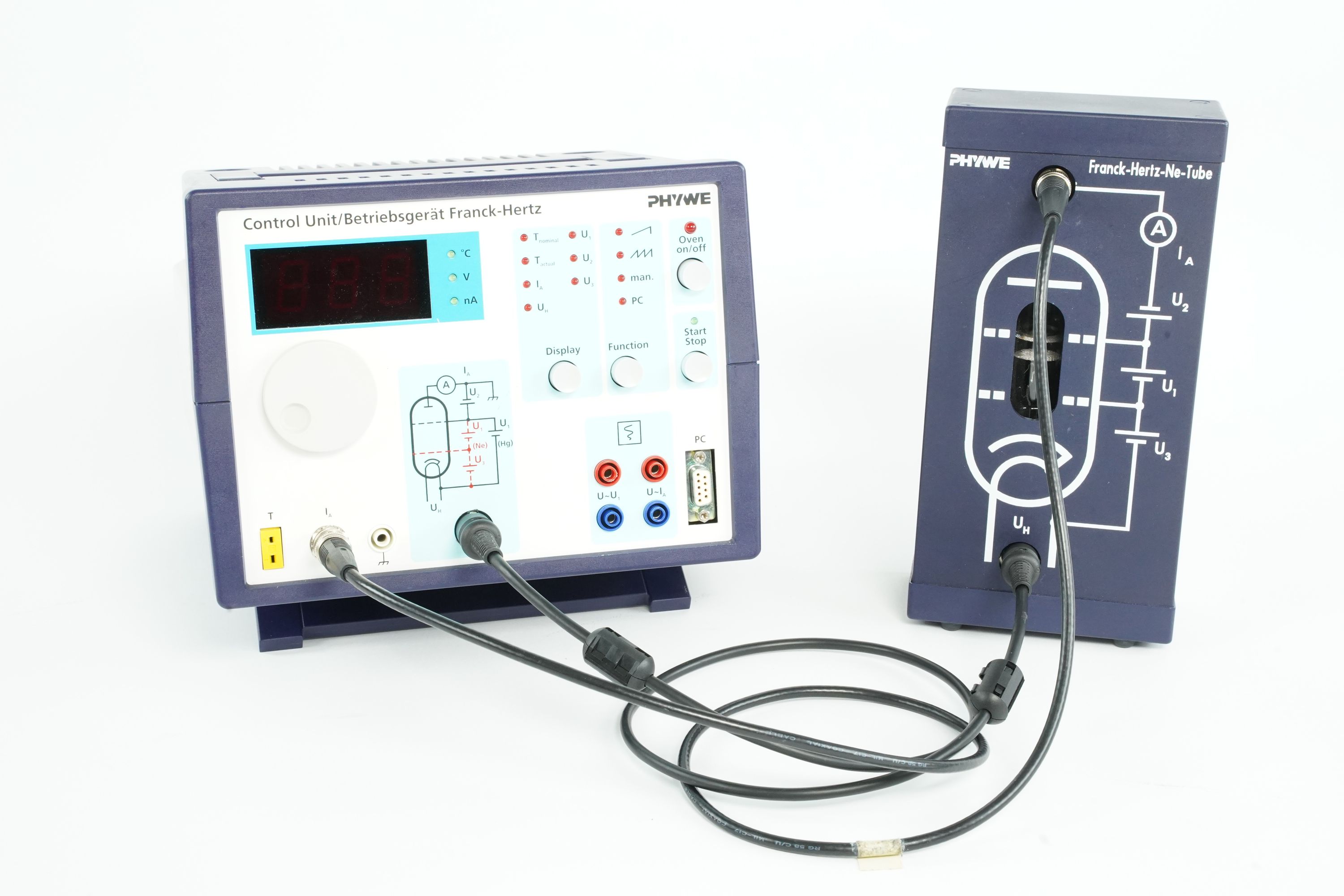
Principle
Electrons are accelerated through a gas of neon atoms. As the electrons collide with the neon atoms, they transfer energy. However, energy transfer only occurs when the electrons have enough energy to excite the neon atoms to a higher energy state. This results in a drop in the current, which is detected as peaks on a voltage-current graph. The neon atoms then emit light as they return to their ground state, producing visible spectral lines. The excitation energy of neon is determined from the distance between the equidistant minima of the electron current in a variable opposing electric field. This experiment provides direct evidence of quantized atomic energy levels.
Benefits
-
Fascinating Nobel Prize experiment (Franck-Hertz 1925)
- Suitable for demonstration and lab courses as well
- Compact neon tube in sturdy casing - ready to use due to short heat-up time
- Control unit compatible to Ne and Hg tube
- Easy-to-use software for control, data acquisition and evaluation
Tasks
- To record the counter current strength I in a Franck-Hertz tube as a function of the anode voltage U.
- To determine the excitation energy E from the positions of the current strength minima or maxima by difference formation.
Learning objectives
- Energy quantum
- Quantum leap
- Electron collision
- Excitation energy
Customers were also interested in...
- Item No.P2510311Franck-Hertz experiment with a Hg-tube